A reaction in COPASI represents a chemical reaction in the model. It consists of substrates, products and optionally modifiers. The substrates are consumed by the reaction, the products are produced by the reaction and modifiers influence the reaction but are neither consumed nor produced by it. A reaction has a kinetic rate law which defines how fast the reaction proceeds.
Adding reactions in COPASI works much like adding compartments or species. To add reactions, navigate to the Reactions branch in the object tree, which is found under Model → Biochemical. This opens a table with six columns:
The the flux is computed automatically when you run a time course simulation.
The simplest way to add a reaction is to enter the chemical equation directly into an empty “Equation” cell in the table. For the syntax see Chemical Equations below. Pressing the return key after typing the equation moves the cursor to the next row, allowing you to quickly add multiple reactions. When you are done entering equations, you “commit” all the reactions at once.
If any of the reaction equations include species that do not yet exist in the model, COPASI will add these species automatically. If there is no compartment yet, COPASI will create one and add all new species to it. If compartments already exist, new species will be added to the first compartment listed in the object tree.
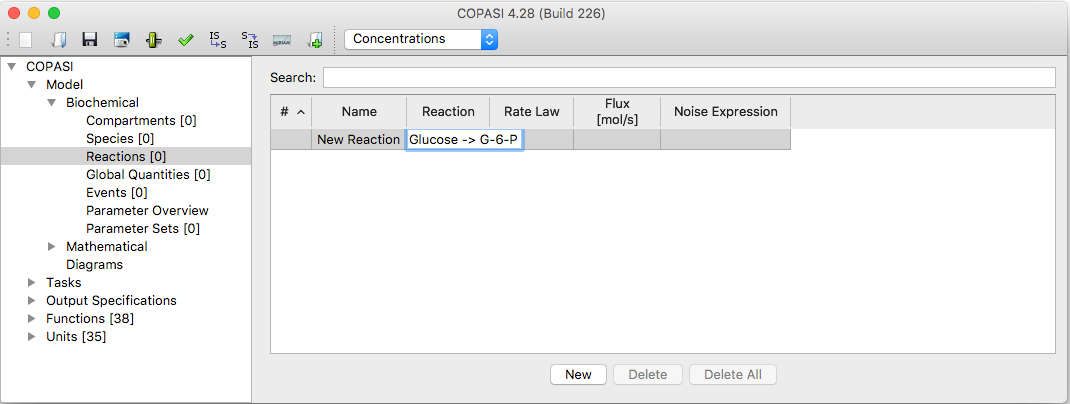 |
| Reaction Table with new Entry |
Each new reaction gets a default kinetic rate law which is irreversible mass action for reactions that contain a
substrate. For reaction that only have a product (e.g. influx into a system) a constant flux kinetic is
chosen (or Mass action (reversible) in case of a reversible reaction).
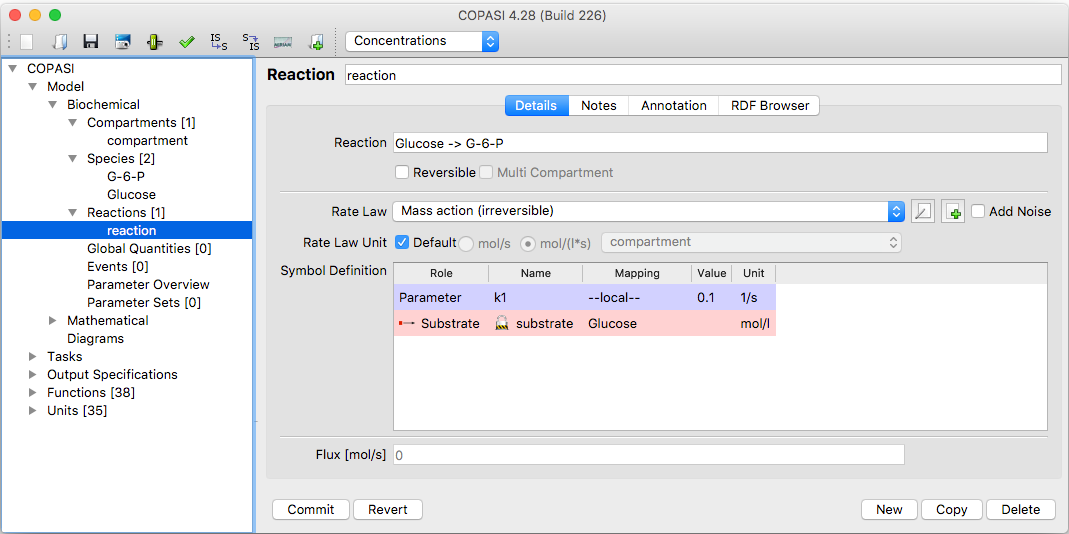 |
| Dialog for changing Reaction Parameters |
<
Double-clicking on a reaction entry in the table opens a dialog where you can edit various parameters for the reaction. Here, you can change the reaction’s name, its chemical equation, and whether or not the reaction is reversible. Altering either the chemical equation or the reversibility affects the list of kinetic rate laws available to be assigned to the reaction. Each kinetic rate law specifies the number of substrates, products, and modifiers it requires, and whether it is suitable only for reversible or irreversible reactions, or both. As a result, only the compatible kinetic functions are shown in the Rate Law dropdown based on your current equation setup. If the kinetic function you want does not exist yet, you can add it with the “New Rate Law” button (see also User Defined Functions). Selecting a kinetic function displays its parameters in the “Symbol Definition” table—each of these parameters is initialized to a default value of 0.1, which you can change by clicking on the cell and entering a new value.
By default, all kinetic function parameters are local; they only apply to the rate law in the selected reaction. In some cases, it may be beneficial to use the same parameter across several reactions. This approach allows you to change the parameter value in a single place, instead of updating it individually in each reaction. Parameters that can be used in multiple reactions are called global quantities in COPASI. For details on creating global quantities, see the Global Quantities section. Suppose you have already defined a global quantity; to use it in a rate law, check the “global” box for the parameter in the Symbol Definition table. COPASI will then let you choose from any global quantities defined in the model. If no global quantities exist, the dropdown will only contain “unknown”—you will first need to define a global quantity and then return to this reaction. If “unknown” is selected, and you leave the dialog, COPASI will automatically revert the parameter to “local.”
Chemical equations in COPASI follow a simple schema. First, list all substrates, separated by a “+” (plus) character, with at least one space before and after each “+”. If you do not include spaces, COPASI will interpret the “+” as part of the species name (which is allowed). After specifying the substrates, indicate whether the reaction is reversible or irreversible by typing either an equals sign (“=”) for reversible, or the character sequence “->” for irreversible. After this, list the products, again separated by “ + “ with spaces. Optionally, you may add a semicolon (;) followed by one or more modifiers, separated by spaces. You may omit either the substrate list or the product list, but at least one must be present.
For example:
A -> BA + B = C; C DFor a detailed explanation of how area scaling and compartment boundaries affect reaction kinetics in models involving multiple compartments, see:
Holzheu, P., Großeholz, R. & Kummer, U. Impact of explicit area scaling on kinetic models involving multiple compartments. BMC Bioinformatics 22, 21 (2021). https://doi.org/10.1186/s12859-020-03913-8